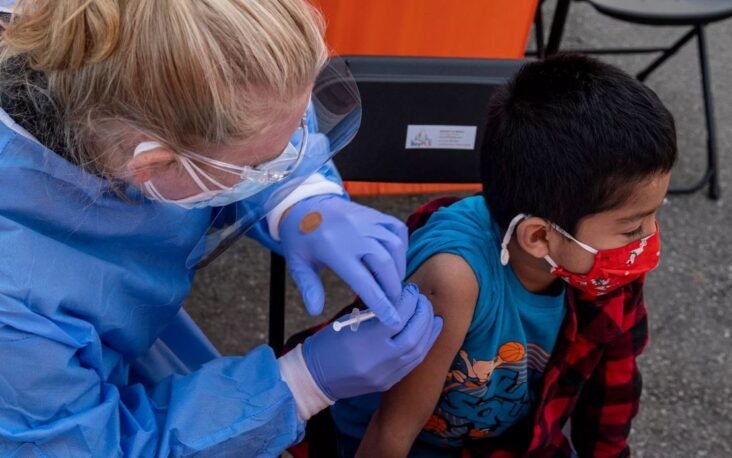
Pfizer-BioNTech is postponing its rolling application to the Food and Drug Administration to expand the use of its two-dose Covid-19 vaccine for children ages 6 months to 4 years.
The move means that vaccines for this age group will not be available in the coming weeks, a setback for parents eager to vaccinate their young children.
Pfizer said on Friday that it will wait for its data on a three-dose series of the vaccine, because it believes three doses “may provide a higher level of protection in this age group.” Data on the third dose is expected in early April, the company said.
Pfizer said in December that two doses didn’t generate a strong enough immune response in its trial of children ages 2 to 4. For young children, Pfizer’s vaccine has a dosage of 3 micrograms. For children ages 5 to 11, the dosage is higher, at 10 micrograms.
Still, the company asked the FDA this month to authorize these first two doses, with a plan to submit additional data in the coming weeks on a third dose. The full vaccination series would be three doses.
The FDA was expected to publish an analysis of the Pfizer data Friday, ahead of an advisory committee meeting next week. The FDA said Friday the meeting has been postponed.
Two people familiar with the FDA’s plans said there had already been a lot of pushback on the agency from outside experts who had concerns that Pfizer’s data wasn’t sufficient. The experts felt, one of the people said, that their concerns were “falling on deaf ears” within the agency.
Federal regulators had initially wanted to begin reviewing the data on two doses of the vaccine while Pfizer continued to gather data on a three-dose regimen.
Regulators believed two doses would provide enough — though less than ideal — protection against the omicron variant of the coronavirus as pediatric cases surged.
However, Dr. Peter Marks, director of the FDA’s Center for Biologics Evaluation and Research, said Friday that after regulators reviewed the company’s two-dose data, they decided they needed to see the three-dose data before considering authorization.
Parents should be “reassured,” Marks said, adding the agency takes its “responsibility for reviewing these vaccines very seriously because we’re parents as well,” he said.
On Tuesday, Pfizer CEO Albert Bourla told CNBC the chances are “very high” the FDA would authorize the vaccine for young children.
“I think that they will be pleased with the data and they will approve,” Bourla told CNBC’s Meg Tirrell, while noting that the regulatory process still has to play out.
Pfizer’s request for authorization for two doses was an unusual request, given that the first two shots didn’t work and there is no guarantee that adding a third dose will be sufficient.
John Moore, a professor of microbiology and immunology at Weill Cornell Medical College, said Pfizer had to make a decision about which dose to study in the clinical trials.
“They have to make the best guess about what dosage to use in young children and they got it wrong, they underdosed,” he said.